Growing up in northern Illinois, I would sometimes walk or run through fields of weeds, fields where many, most, or all of the weeds were goldenrods. After some of those forays, I noticed a few blue-black insect larvae on my jeans. I had no idea what they were, and it was immediately obvious that they were just accidental passengers, easily removed and nothing for me to worry about.
I later learned that they were the larvae of Trirhabda beetles, a genus of leaf beetles (Chrysomelidae) whose species each feed on a small range of host plants. At least two species, T. canadensis and T. virgata, specialize on goldenrods, both as larvae and as adults.
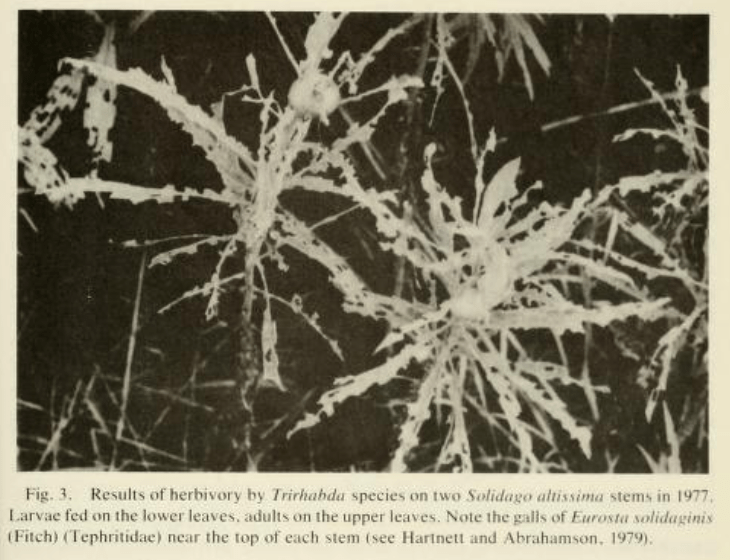
The beetles cling to the surface of the plant and chew up the leaf tissue. Each beetle can eat a fair amount of leaf material in its lifetime, and sometimes, there are a large number of beetles, each roughly a centimeter long. After most of season of feeding on goldenrods, sometimes there isn’t much leaf tissue remaining (see photo above).
One might think that these beetles, exposed on the plant, would be easy pickings for predators, and some predators and parasites do attack them. But the beetles smell like goldenrods, a distinctive odor that results from an array of chemicals in the plant tissues, chemicals other than the content that provides nutrition for the beetles. When the late Robert Whittaker was a professor at Cornell, he tried to feed a pet lizard some Trirhabda beetles. Though the lizard was quick to devour mealworm larvae, it wanted nothing to do with the goldenrod beetles. Their chemical content, taken from their goldenrod food, seemed to provide some degree of defense (more on this in another post).
I don’t have a picture of Trirhabda beetles, but you can easily find some on line (for example: https://en.wikipedia.org/wiki/Trirhabda_canadensis).
Ann Herzig performed some elegant experiments with Trirhabda beetles and goldenrods in and around Ithaca, New York. She planted goldenrods on the roofs of some buildings on the Cornell campus because the buildings were a long way from the fields of goldernrods around the city. In various plots, she produced different amounts of beetle-like damage to see whether the beetles responded to the leaf damage they encountered. The beetles found these plants (clearly, their dispersal abilities were considerable) and they were more likely to stay on plants with low levels of damage. At least some of the females that arrived on her test plots had already mated, and they laid more eggs on plots with low damage than on plots with high damage.
When she set up other plots around Ithaca with different levels of damage, beetles tended to leave plots with high damage, and they tended to stay on plots with low damage. If she put a dab of paint on the back of the beetle, gluing the forewings (elytra) together, they couldn’t fly (this was a brilliant experimental manipulation). The painted beetles stuck around longer than other beetles, which might seem like an obvious result, but she couldn’t be sure of their main mode of dispersal until she prevented them from flying.
So she showed (among other things) that the beetles can find goldenrods that are far away, can detect levels of damage on the goldenrods, and will fly elsewhere if the plants are already heavily consumed. These beetles are well adapted for the exploitation of goldenrods.
But for various reasons (most of which I don’t know), the numbers of beetles fluctuate from year to year, so the goldenrods are not always slammed by these herbivores. The plants and insects manage to coexist over a fairly large portion of North America. Is their coexistence the result of coevolution? Seems like a reasonable hypothesis.
One last thought: goldenrod ball galls can reduce the weight of seeds and the number of rhizomes produced by an infested goldenrod plant. I suspect that Trirhabda, when they are abundant, can have an even greater effect on goldenrod growth. Seems like another reasonable hypothesis.
Further reading
Ann L. Herzig 1995 Effects of Population Density on Long-Distance Dispersal in the Goldenrod Beetle Trirhabda virgata. Ecology 76: 2044-2054
Ann L. Herzig 1996 Colonization of host patches following long-distance dispersal by a goldenrod beetle, Trirhabda virgata. Ecological Entomology 21: 344-351
Owen D.V. Sholes. 1981 Herbivory by species of Trirhabda (Coleoptera: Chrysomelidae) on Solidago altissima(Asteraceae): variation between years. Proceedings of the Entomological Society of Washington (D.C.) 83: 274–282
David C Hartnett and Warren G Abrahamson 1979 The Effects of Stem Gall Insects on Life History Patterns in Solidago canadensis. Ecology 60: 910-917